Two cyanine derivatives, Cyanine 3 and Cyanine 5, which differ in structure simply by the number of carbons in the conjugated poly-ene linkage, are joined by the closely related analogues, Cyanine 3.5 and Cyanine 5.5, and are available as phosphoramidites. Cyanine dyes are normally added once at the 5’-terminus and the MMT group should be removed on the synthesizer. The absorbance of the MMT cation (yellow) is noticeably different from the DMT cation (orange), and so, absorbance-based trityl monitors will detect it incorrectly as a low coupling. On the other hand, conductivity detectors will interpret the release more correctly. Cyanine dye phosphoramidites have also been used successfully adjacent to the 3’-terminus. Cyanine 3 and Cyanine 5 supports are also offered to allow simpler production of 3’ cyanine dye-labeled oligonucleotides.
Deprotection of oligos containing Cyanine dyes may be carried out with ammonium hydroxide at room temperature, regardless of the base protecting groups on the monomers used. If there is a need to use ammonium hydroxide at elevated temperature, Cyanine 3 and Cyanine 3.5 are more stable than Cyanine 5 and Cyanine 5.5. However, it is always prudent to use monomers with base labile protecting groups to limit the exposure time to 2 hours or less at 65°C during deprotection.
To better address applications in near-infrared (NIR) imaging, Glen Research is offering a water soluble Disulfo-Cyanine 7 azide that can be easily conjugated to DNA and RNA through standard click chemistry. This long wavelength dye offers the benefits of improved solubility, reduced aggregation, and improved stability in the near-infrared spectrum along with the convenience of click chemistry.
The spectral characteristics of these dyes are detailed here.

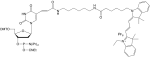


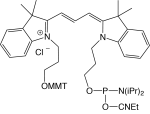
1-[3-(4-monomethoxytrityloxy)propyl]-1'-[3-[(2-cyanoethyl)-(N,N-diisopropyl)phosphoramidityl]propyl]-3,3,3',3'-tetramethylindocarbocyanine chloride
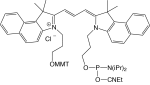
1-[3-(4-monomethoxytrityloxy)propyl]-1'-[3-[(2-cyanoethyl)-(N,N-diisopropyl)phosphoramidityl]propyl]-3,3,3',3'-tetramethyl-4,5-benzindocarbocyanine chloride
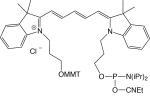
1-[3-(4-monomethoxytrityloxy)propyl]-1'-[3-[(2-cyanoethyl)-(N,N-diisopropylphosphoramidityl]propyl]-3,3,3',3'-tetramethylindodicarbocyanine chloride
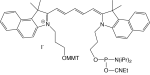
1-[3-(4-monomethoxytrityloxy)propyl]-1'-[3-[(2-cyanoethyl)-(N,N-diisopropyl)phosphoramidityl]propyl]-3,3,3',3'-tetramethyl-4,5-benzindodicarbocyanine iodide

1-[3-(4-monomethoxytrityloxy)propyl]-3,3,3',3'-tetramethylindocarbocyanine chloride-1'-propyl-3-O-succinoyl-long chain alkylamino-CPG

1-[3-(4-monomethoxytrityloxy)propyl]-3,3,3',3'-tetramethylindodicarbocyanine chloride-1'-propyl-3-O-succinoyl-long chain alkylamino-CPG