Glen Report 19.11: Modified RNA Phosphoramidites Useful in siRNA Research and Biologically Significant 1-Methyl-adenosine
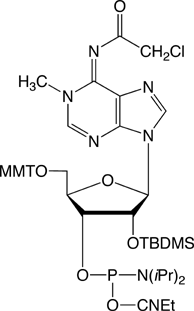
In all 20 years of Glen Research’s existence, we have heard prediction after prediction that RNA synthesis was about to enjoy explosive growth. Although activity certainly increased slightly over the years thanks to studies on RNA structure and function relative to RNA viral activity, t-RNA and ribozymes (among others) growth could never have been defined as explosive. Finally, these predictions seem to be coming to fruition thanks to the advent of siRNA. Currently, researchers seem to be happy with the ready availability of unmodified siRNA but we predict that interest will continue to rise in the ability to use base, sugar and phosphate modifications to optimize siRNA activity. In this short article, we will focus on base modifications that are potentially useful in siRNA research. In the following article on Page 3, we will highlight a modified RNA base with significant biological interest and potential: 1-Methyl-Adenosine (m1A), the phosphoramidite of which is shown in Figure 1 on this page.
A quick glance through our catalog or a browse through our web site quickly reveals a significant selection of base modified and minor RNA monomers available from stock. Here is the current list:
- Pyrrolo-Cytidine (10-3017)
- Inosine (10-3040)
- 5-Methyl-Uridine (ribo version of Thymidine) (10-3050)
- 4-Thio-Uridine (10-3052)
- 5-Methyl-Cytidine (10-3064)
- 2-Aminopurine Ribonucleoside (10-3070)
- 6-Thio-Guanosine (10-3072)
- 2,6-Diaminopurine Ribonucleoside (2-Amino-Adenosine) (10-3084) has been discontinued see 2,6-Diaminopurine-TOM-CE Phosphoramidite (10-3085)
- 5-Bromo-Uridine (10-3090)
- 5-Iodo-Uridine (10-3091)
- Amino-Modifier C6-U (10-3039)
- rSpacer (10-3914) has been discontinued
We are happy to add to this list of RNA phosphoramidites the following naturally-occurring or modified bases:
- 7-Deaza-Adenosine (10-3001) has been discontinued
- 8-Aza-7-deaza-Adenosine (10-3083) has been discontinued
- Zebularine (10-3011)
- Pyridin-2-one Ribonucleoside (10-3012)has been discontinued
- Nebularine (Purine Ribonucleoside) (10-3041) has been discontinued
- PseudoUridine (10-3055)
The structures of these RNA phosphoramidites are shown in Figure 2 and the following is a very brief introduction to each of these monomers.
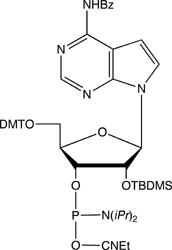
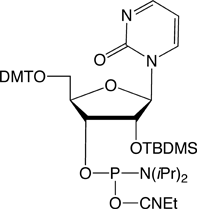
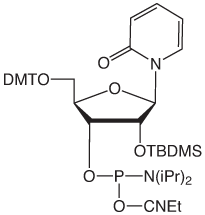



Adenosine Analogues
7-Deaza-Adenosine is lacking nitrogen at the 7-position, which is replaced by carbon. The N7 position in adenosine takes part in non-Watson and Crick hydrogen bonding, which may be relevant to RNA folding and subsequent activity. This Adenosine analogue is also known as Tubercidin. 8-Aza-7-deaza-Adenosine is an isomer of Adenosine with virtually identical electron density. Again, the N7 nitrogen is not available for hydrogen bonding. Nebularine or Purine Nucleoside can be viewed as an Adenosine derivative that is lacking the exocyclic amino group. This molecule allows researchers to determine the relevance of the exocyclic amine of Adenosine to RNA structure and function. If functionality is lost after substitution of Nebularine for Adenosine, then hydrogen bonding involving the exocyclic amine can be inferred, while retention of functionality infers the opposite.
Cytidine Analogues
Ribozyme activity is substantially affected by the substitution of modified pyrimidine bases. Zebularine (pyrimidin-2-one ribonucleoside) may be regarded as a Cytidine derivative lacking the exocyclic amino group. Zebularine and Pyridin-2-one Ribonucleoside, the 3-deaza analogue of Zebularine, are prime candidates for use in evaluating ribozyme activity and function. It should be noted that Zebularine is mildly fluorescent, absorbing at 298nm and emitting at 367nm.
PseudoUridine
PseudoUridine is one of the most common modified nucleosides found in RNA. The availability of a phosphoramidite will allow detailed research into the effects of this modified base on RNA structure and activity.
- Glen Report 19.11: Modified RNA Phosphoramidites Useful in siRNA Research and Biologically Significant 1-Methyl-adenosine
- Glen Report 19.12: 1-Methyl-Adenine in Nucleic Acids
- Glen Report 19.13: Technical Brief - Precautions During Packaging of Phosphoramidites and Transfer into Alternative Vials
- Glen Report 19.14: Microarrays, Nanotechnology and Beyond
- Glen Report 19.15: 5’-Hexynyl Phosphoramidite – Conjugation with a Click
- Glen Report 19.16: Quenched Autoligation (QUAL) Probes
- Glen Report 19.17: Selenium Derivatization of Nucleic Acids for X-ray Crystal Structure Determination
- Glen Report 19.18: Large Scale Synthesis Update
- Glen Report 19.19: NHS-Carboxy-dT – Expanding the NHS Ester Phosphoramidite Repertoire

