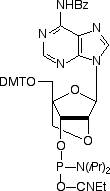
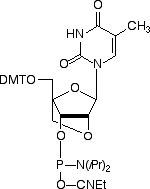
Locked Nucleic Acid (LNA) was first described by Wengel and co-workers in 19981 as a novel class of conformationally restricted oligonucleotide analogues. LNA is a bicyclic nucleic acid where a ribonucleoside is linked between the 2’-oxygen and the 4’-carbon atoms with a methylene unit. The structures are detailed in Figure 1. Under licence from Exiqon A/S (Denmark), and in association with Link Technologies Ltd (Scotland), we are now able to offer the four standard LNA phosphoramidites.
| Bz-A-LNA | 5-Me-Bz-C-LNA | dmf-G-LNA | T-LNA |
 |
 |
 |
 |
Oligonucleotides containing LNA exhibit unprecedented thermal stabilities towards complementary DNA and RNA2, which allows excellent mismatch discrimination. In fact the high binding affinity of LNA oligos allows for the use of short probes in, for example, SNP genotyping3, allele specific PCR and mRNA sample preparation. In fact, LNA is recommended for use in any hybridization assay that requires high specificity and/or reproducibility, e.g., dual labelled probes, in situ hybridization probes, molecular beacons and PCR primers. Furthermore, LNA offers the possibility to adjust Tm values of primers and probes in multiplex assays.
As a result of these significant characteristics, the use of LNA-modified oligos in antisense drug development is now coming under investigation4, and recently the therapeutic potential of LNA has been reviewed.5
In general, LNA oligonucleotides can be synthesized by standard phosphoramidite chemistry using automated DNA synthesizers. The phosphoramidites can be dissolved in anhydrous acetonitrile to standard concentrations, except for the 5-Me-C variant which requires the use of a 25% THF/acetonitrile solution. They are more sterically hindered compared to standard DNA phosphoramidites and therefore require a longer coupling time. 180 seconds and 250 seconds coupling times are recommended for ABI and Expedite synthesizers, respectively.
The oxidation of the phosphite after LNA coupling is slower compared to the similar DNA phosphite, and therefore a longer oxidation time is suggested. Using standard iodine oxidation procedures, 45 seconds has been found to be the optimal oxidation time on both ABI and Expedite instruments. LNA-containing oligo-nucleotides are deprotected following standard protocols. It is, however, advisable to avoid the use of methylamine when deprotecting oligos containing Me-Bz-C-LNA since this can result in introduction of an N4-methyl modification.
LNA-containing oligonucleotides can be purified and analyzed using the same methods employed for standard DNA. LNA can be mixed with DNA and RNA, as well as other nucleic acid analogues, modifiers and labels. LNA oligonucleotides are water soluble, and can be separated by gel electrophoresis and precipitated by ethanol.
Locked-nucleic Acid (LNA) phosphoramidites are protected by EP Pat No. 1013661, US Pat No. 6,268,490 and foreign applications and patents owned by Exiqon A/S. Products are made and sold under a license from Exiqon A/S. Products are for research purposes only. Products may not be used for diagnostic, clinical, commercial or other use, including use in humans. There is no implied license for commercial use, including contract research, with respect to the products and a license must be obtained directly from Exiqon A/S for such use.