Glen Report 16.25: Trimer (Codon) Phosphoramidites Simplify Library Preparation
Oligonucleotide-directed mutagenesis is probably the most popular approach for the preparation of proteins with variations at specific sites. This protein engineering technique uses oligonucleotides of mixed sequences to generate libraries of proteins for screening potential improvements in specific biological function. It is certainly possible to produce the mixed oligonucleotide sequences by opening the synthesis columns, splitting the supports, and recombining the supports after coupling. This procedure is surely labor-intensive and coupling efficiency is always affected by the splitting and recombination process. The technique is also limited in that the complexity desired may be greater than the number of particles of support in the columns. Another technique is to use mixtures of monomers to generate codon mixtures but the degeneracy of the genetic code guarantees that redundancies and stop codons will be generated. Mutagenesis generating substoichiometric amounts of codons at specific positions has been described1,2 using a mixture of trimer and monomer phosphoramidites. A further refinement of this strategy has been described3 using two sets of monomers, one set with 5'-DMT protection and one set with base-labile 5'-Fmoc protected monomers.
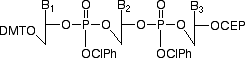
General Structure of Trimer Phosphoramidites,where B=Abz, Cbz, Gibu, T
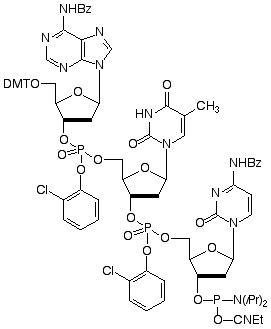
Detailed Structure of ATC
Figure 1: Structure of trimer phosphoramidites
In principle, the simplest approach for oligonucleotide-directed mutagenesis would be the use of trimer phosphoramidites. Of the 64 possible combinations of codons, only 20 codons would be required to cover the 20 amino acids, although, in practice, several codons will likely be duplicated depending on the organism. Several reports describing4-6 the synthesis of trimer phosphoramidites have been published. We prefer the approach described7-9 by Kayushin et al and our trimers use their protection scheme.
Quality control of trimer phosphoramidites is very challenging. We normally use RP HPLC for purity and identity determination of our regular phosphoramidites. However, trimer phosphoramidites have chiral centers at all three phosphorus positions. There are, therefore, 23 = 8 diastereomers in each phosphoramidite, which are at least partially separated on RP HPLC, rendering the technique questionable for purity and identity determination. There is also the concern that the sequence of the trimers has to be verified. For example, CAT coding for histidine, has to be differentiated from TAC, coding for tyrosine. These two trimers have virtually identical lipophilicity and their identity cannot be clearly confirmed by HPLC. This problem has been solved10 using HPLC electrospray mass spectrometric analysis of the trimers, which provides data confirming molecular weight and sequence.
In Table 1, the trimers, their coding amino acid and their reaction factor (RF) are listed. The reaction factor is critical since the trimers will likely be mixed and they have differing reactivity in the coupling reaction. RF for AAC is 1.0 and for TAC is 1.6. Therefore, 1.6 equivalents of TAC are needed for every 1.0 equivalent of AAC for equal coupling. Mixtures can easily be made using equimolar solutions or the molecular weight of each trimer has to be used to generate the appropriate weights of each trimer to use if mixing by weight. An example of the preparation of a mixture of all 20 trimers is shown in the right column of Table 1 and completed in the footnotes.
| Trimer | Amino Acid | Abbreviation | MW | RF | MWxRF | mg/10µmol (adjusted for RF) |
|---|---|---|---|---|---|---|
| AAA | Lys | K | 1911.5 | 1.1 | 2102.65 | 21.0 (11) |
| AAC | Asn | N | 1887.5 | 1.0 | 1887.50 | 18.9 (10) |
| ACT | Thr | T | 1774.5 | 1.3 | 2306.85 | 23.1 (13) |
| ATC | Ile | I | 1774.5 | 1.2 | 2129.40 | 21.3 (12) |
| ATG | Met | M | 1780.5 | 1.3 | 2314.65 | 23.1 (13) |
| CAG | Gln | Q | 1869.5 | 2.0 | 3739.00 | 37.4 (20) |
| CAT | His | H | 1774.5 | 1.9 | 3371.55 | 33.7 (19) |
| CCG | Pro | P | 1845.5 | 1.8 | 3321.90 | 33.2 (18) |
| CGT | Arg | R | 1756.5 | 1.1 | 1932.15 | 19.3 (11) |
| CTG | Leu | L | 1756.5 | 1.2 | 2107.80 | 21.1 (12) |
| GAA | Glu | E | 1893.5 | 1.9 | 3597.65 | 36.0 (19) |
| GAC | Asp | D | 1869.5 | 1.3 | 2430.35 | 24.3 (13) |
| GCT | Ala | A | 1756.5 | 1.5 | 2634.75 | 26.3 (15) |
| GGT | Gly | G | 1762.5 | 1.1 | 1938.75 | 19.4 (11) |
| GTT | Val | V | 1667.5 | 1.9 | 3168.25 | 31.7 (19) |
| TAC | Tyr | Y | 1774.5 | 1.6 | 2839.20 | 28.4 (16) |
| TCT | Ser | S | 1661.4 | 1.3 | 2159.82 | 21.6 (13) |
| TGC | Cys | C | 1756.5 | 1.5 | 2634.75 | 26.3 (15) |
| TGG | Trp | W | 1762.5 | 2.4 | 4230.00 | 42.3 (24) |
| TTC | Phe | F | 1661.4 | 1.3 | 2159.82 | 21.6 (13) |
| =530.0 mg | ||||||
Example of Preparation of Trimer Mixture
Prepare 592.6 mg of the trimer mix, taking the amount (mg) for each trimer from the right column. Dissolve the trimer mix in dichloromethane (highest grade possible; acid-free). Evaporate to dryness to produce a homogenous mixture of all 20 trimers.
Example of Preparation of Trimer Mixture for the Synthesizer
Dissolve 592.6 mg, which is equivalent to 20X10 µmoles (normalized for RF) of the trimer mix in 2.0 mL of acetonitrile-dichloromethane mixture, 1:3 v/v to produce a 0.10N solution of trimers, ready for use in a synthesizer.
All of the trimers are now available individually so that researchers can prepare custom mixtures. A mixture of all 20 trimers designed to produce equal coupling of all 20 is also available. If you require custom production of a specific mixture, please e-mail support@glenresearch.com for a quotation and projected delivery.
References
- J. Sondek and D. Shortle, Proc Natl Acad Sci U S A, 1992, 89, 3581-3585.
- P. Gaytan, J. Yanez, F. Sanchez, H. Mackie, and X. Soberon, Chem Biol, 1998, 5, 519-527.
- P. Gaytan, J. Yanez, F. Sanchez, and X. Soberon, Nucleic Acids Res, 2001, 29, E9.
- B. Virnekas, L.M. Ge, A. Pluckthun, K.C. Schneider, G. Wellnhofer, and S.E. Moroney, Nucleic Acids Research, 1994, 22, 5600-5607.
- M.H. Lyttle, E.W. Napolitano, B.L. Calio, and L.M. Kauvar, Biotechniques, 1995, 19, 274.
- A. Ono, A. Matsuda, J. Zhao, and D.V. Santi, Nucleic Acids Research, 1995, 23, 4677-4682.
- A.L. Kayushin, M.D. Korosteleva, A.I. Miroshnikov, W. Kosch, D. Zubov, and N. Piel, Nucleic Acids Research, 1996, 24, 3748-3755.
- A. Kayushin, et al., Nucleos Nucleot, 1999, 18, 1531-1533.
- A. Kayushin, M. Korosteleva, and A. Miroshnikov, Nucleos Nucleot Nucleic Acids, 2000, 19, 1967-1976.
- T. Mauriala, S. Auriola, A. Azhayev, A. Kayushin, M. Korosteleva, and A. Miroshnikov, J. Pharmaceutical and Biomedical Analysis, In Press.
Product Information
- Glen Report 16.21: Thymine dimers - DNA Lesions Induced By Sunlight CIS-SYN Thymine Dimer Phosphoramidite Now Available
- Glen Report 16.22: EraGen BioSciences Technology (courtesy of EraGen Biosciences)
- Glen Report 16.23: Activators, Columns and Plates
- Glen Report 16.24: Locked Nucleic Acid (LNA™) Phosphoramidites
- Glen Report 16.25: Trimer (Codon) Phosphoramidites Simplify Library Preparation
- Glen Report 16.26: Still More New Products
- Glen Report 16.27: New Epoch Products - 5'-Aldehyde-Modifier C2 Phosphoramidite and Gig Harbor Green™ Phosphoramidite

