Glen Report 4-13: RNA Synthesis - 2'-O-Methyl Analogues
Oligonucleotides consisting of 2'-O-methyl-ribonucleotides (2'-OMe RNA) are arousing considerable interest as antisense probes since 2'-OMe RNA forms a stable duplex with RNA, while being resistant to RNase H activity1. At the same time , the 2'-OMe RNA is fully resistant to various RNase and DNase activities2. Of specific interest to researchers is the use of 2'-OMe RNA as antisense probes for studying site-specific inhibition of pre-mRNA splicing. The preparation of mixed oligoribonucleotides containing 2'-OMe ribonucleotides at specific points has been described3, along with their significance in the study of RNA - protein interactions and their application to the synthesis of stable ribozymes.
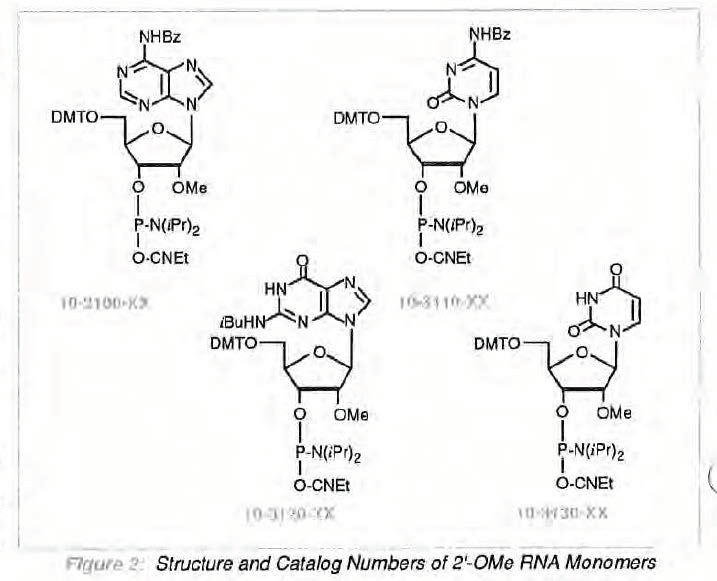
As with all seemingly exciting developments, there are significant hurdles to overcome before the full potential of these techniques may be realized. The production of the CE Phosphoramidite monomers for use in 2'-OMe RNA synthesis is very difficult, With the G derivative (and to only a slightly lesser extent the A derivative) being particularly challenging. For a flavor of the synthesis of the purine monomers, check out the most up-to -date description4 in which the G monomer is produced in a tortuous 12-step synthesis and the A monomer in a mere 7 steps! The pyrimidine monomers are, at least, slightly easier to produce5.
The difficulties in synthesis of these monomers do not, fortunately, extend to the use of the monomers . Synthesis of 2'-OMe RNA proceeds in a manner analogous to RNA synthesis with a coupling time of 15 minutes. The deprotection steps are identical to those used for DNA synthesis since the 2'-OMe groups remain intact. Also, since the sequences are resistant to RNase hydrolysis, purification can be effected by the techniques normal to DNA synthesis, without the need for sterile conditions.
Because of the long and difficult synthesis of 2'-OMe RNA monomers, we are unable to guarantee that stocks will be sufficient to meet projected needs . However, we do guarantee that we will not compromise on quality and performance of monomers.
References
(1) A. Mayeda, Y. Hayase, H, Inoue, E.. Ohtsuka , and Y. Ohshima , J, Biochem., 1990, 108 , 399.
(2) B.S. Sproat, A.I. Lamond, B. Beijer, P. Neuner, and U. Ryder, Nucleic Acids Res., 1989 , 17, 3373.
(3) 8. Beijer, I. Sulston, 8.S. Sproat, P. Rider , A.I. Lamond , and P. Neuner, Nuc/eioAcids Res., 1990, 18, 5143-5151. (
(4) B.S. Sproat, B. Beijer, and A. Jrabarren, Nucleic Acids Res., 1990, 18, 41.
(5) A. Nyilas and J. Chattopadhyaya , Acta Chem. Scand ., 1986, B40, 826.
Product Information
- Glen Report 4-11: Introduction
- Glen Report 4-12: RNA Synthesis - Options for 2'-OH Protection
- Glen Report 4-13: RNA Synthesis - 2'-O-Methyl Analogues
- Glen Report 4-14: Literature Review
- Glen Report 4-15: Biotin Phosphoramidites
- Glen Report 4-16: Sulfurization of Oligonucleotides
- Glen Report 4-17: Deoxynucleoside Analogues

