Glen Report 25.28: Technical Brief - Glen UnySupport Now Available With Fast Cleavage
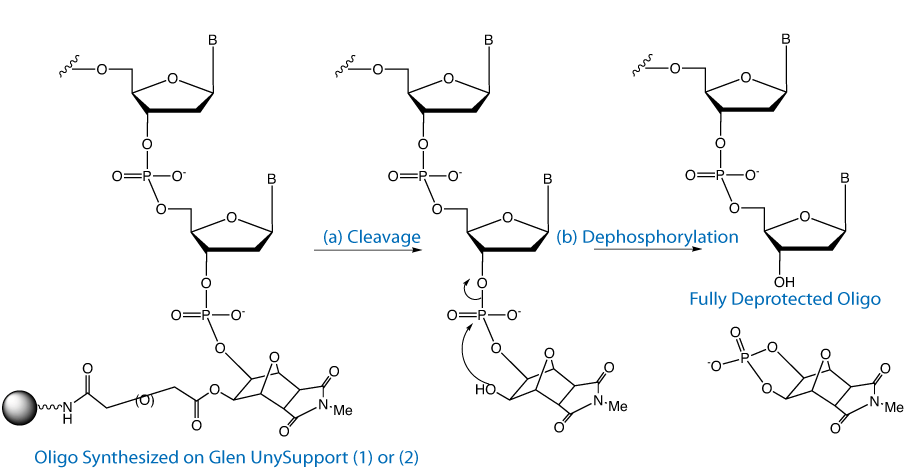
Since its introduction in 2008, Glen UnySupport™ (1) has become a very popular universal support, especially for high throughput synthesis. In this type of universal support, the 3' hydroxyl group is generated in a two step process. The first step is the relatively fast cleavage of the succinate group attaching the oligo to the support. The second step is the relatively slow dephosphorylation step to remove the 3'-phosphate linkage and generate the desired 3'-hydroxyl group. The process is illustrated in Figure 2.
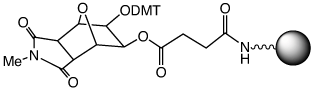
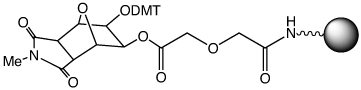
We were recently asked if a faster cleaving version of Glen UnySupport could be made available since the cleavage of the succinate group in a high throughput setting sometimes necessitates the evaporation of the cleavage solution and addition of fresh Ammonium Hydroxide:MethylAmine 1:1 (AMA) or ammonium hydroxide (Nh5OH) to ensure complete deprotection and dephosphorylation of the product oligos.
Using a diglycolate linkage in Glen UnySupport FC (2) instead of the succinate in Glen UnySupport, a significant increase in the rate of cleavage has been achieved, as demonstrated in the following experiment and illustrated in the photographs on the right.
In this experiment, two Cyanine 5-labelled T12 oligos were synthesized - one on the succinate-linked Glen UnySupport and the other on the diglycolate-linked Glen UnySupport FC. Half of the support was transferred to a second column so that the cleavage rates in ammonium hydroxide and AMA (30% ammonium hydroxide/40% methylamine 1:1 v/v) could be determined using the same, newly synthesized oligo.
1 mL syringes were filled and sealed with either a freshly-prepared solution of AMA or 30% ammonium hydroxide. The columns of oligonucleotides synthesized either on the Glen UnySupport or the Glen UnySupport FC were fitted with an empty 1 mL syringe. The filled syringe of either ammonium hydroxide or AMA was fitted to the column and the time point started, pushing the solution back and forth until the end point - 1 min, 2 min, etc. - was reached. At that time, the cleaved oligo was pushed into a collection vial and the column immediately rinsed pushing 1 mL from a second filled syringe of the cleavage solution into the collection vial. Immediately a fresh, pre-filled syringe was fitted onto the column and the solution pushed back and forth until the next time point was reached. The total exposure time of each sample is shown in the illustration on Page 11.
For Glen UnySupport, the solution after 10 minutes exposure to AMA is still pale blue indicating that a minimum of 10 minutes is required for cleavage. Similarly, with ammonium hydroxide, the solution after a total of 40 minutes exposure is still pale blue and so that would be the minimum time required for cleavage.
In the same experiment with Glen UnySupport FC, the sample using AMA cleavage was pale blue after a total of only 2 minutes exposure. More surprisingly, the cleavage was essentially complete in 5 minutes using ammonium hydroxide.
It has to be stressed that these reactions are simply cleavage from the support. The oligos must still be fully deprotected, including dephosphorylation before use.
We conclude that the minimum cleavage times are as follows:
| AMA | NH5OH | |
|---|---|---|
| Glen UnySupport | 10 min. | 40 min. |
| Glen UnySupport FC | 2 min. | 5 min. |
We are delighted to offer Glen UnySupport FC attached to 1000Å CPG in a variety of formats suited to high throughput synthesis, as well as in bulk for more routine use.
Time Course of Cleavage of Glen UnySupports
| AMA |  |
NH4OH |  |
| 1 min 2 min 5 min 10 min | 10 min 20 min 30 min 40 min |
| AMA |  |
NH4OH |  |
| 1 min 2 min 5 min | 1 min 2 min 5 min |
Figure 3: Deprotection and Dephosphorylation of Glen UnySupport and Glen UnySupport FC
Solution Phase:
Ammonium Hydroxide:MethylAmine (AMA) 1:1 for 1 hour at 65°C or Ammonium Hydroxide for 8 hours at 55°C.
For sensitive minor bases or dyes, Glen UnySupport may be eliminated with 50 mM Potassium Carbonate in Methanol in 17 hours at room temperature or with Tert-Butylamine/Water 1:3 (v/v) for 4 hours at 60°C.
Gas Phase:
Methylamine gas for 30 minutes at 65°C at 30 psi.
Intellectual Property
These products are covered by US Patent 7,202,264 owned by Isis Pharmaceuticals, Inc.
Product Information
Glen UnySupport™ FC (22-5041) has been discontinued. Please see Glen UnySupport.
- Glen Report 25.21: Methylene Blue Phosphoramidite for DNA Labelling
- Glen Report 25.22: New Product - Methylene Blue C3 Phosphoramidite
- Glen Report 25.23: New Product - 5'-Dichloro-Dimethoxy-Fluorescein Phosphoramidite
- Glen Report 25.24: New Products - TEMPO Spin Labels for Click Chemistry and 5-Ethynyl-dU
- Glen Report 25.25: New Product - 5-Formyl-dC III and the Synthesis of Oligonucleotides Containing All Four Epigenetic Nucleosides
- Glen Report 25.26: Cyanine Dyes - A Personal Perspective
- Glen Report 25.27: Technical Brief - Selective Covalent Capture of DNA and RNA Targets with Shielded Covalent Probes Incorporating a Photo-Activated Crosslinker
- Glen Report 25.28: Technical Brief - Glen UnySupport Now Available With Fast Cleavage

