Glen Report 24.17: New Product - 5'-Stearyl Phosphoramidite - An Alternative Lipophilic Carrier
Synthetic oligonucleotides that have been modified chemically to achieve certain biological characteristics have found uses in therapeutic development. For example, antisense oligonucleotides, siRNA, aptamers and miRNA have all been found to have qualities that justify pharmaceutical development. As always, the challenge of such development has been to deliver the oligos effectively to the desired cellular targets. A variety of delivery strategies is currently in use but a favorite has been simple modification of the oligonucleotide at the 5' terminus with a lipophilic and non-toxic carrier.
For example, cholesterol-conjugated siRNAs have been shown to silence gene expression in vivo. In a review article,1 the authors described the preparation and use of siRNA modified with a variety of lipophilic carriers. They demonstrated that bile acids and long-chain fatty acid conjugates, in addition to cholesterol, help with siRNA delivery into cells with resulting silencing of gene expression. The authors note that efficient and selective uptake of these siRNA conjugates depends on interactions with lipoprotein particles, lipoprotein receptors and transmembrane proteins.1
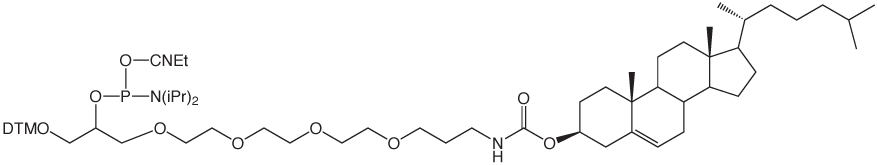
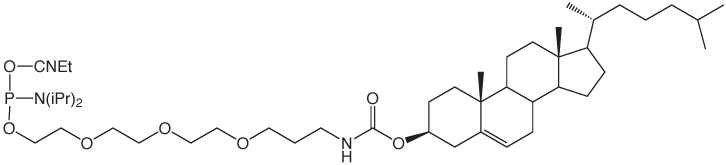
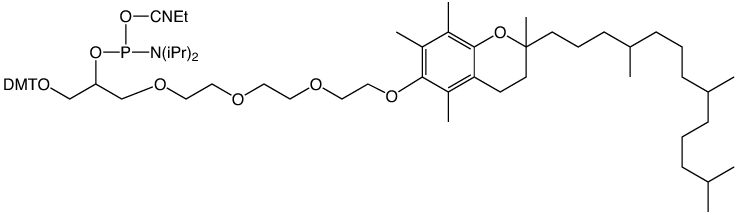
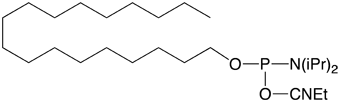
Glen Research has supported these activities for several years with cholesterol and a-tocopherol carriers (1-3). We now offer a simple C18 lipid as an economical and effective carrier molecule.2 We envisage that 5'-Stearyl Phosphoramidite (4) will become a favored lipophilic carrier for experimentation with synthetic oligonucleotides.
5'-Stearyl Phosphoramidite is not fully soluble in acetonitrile and must be dissolved in a mixture of acetonitrile/dichloromethane (1:3). Otherwise the coupling reaction at the 5' terminus is standard with a 3 minute coupling time recommended.
5'-Stearyl oligonucleotides can be purified using standard procedures on RP cartridges, such as Glen-Pak™ cartridges, as well as standard chromatographic techniques.
References
- C. Wolfrum, et al., Nat Biotechnol, 2007, 25, 1149-57.
- H. Liu, B. Kwong, and D.J. Irvine, Angew Chem Int Ed Engl, 2011, 50, 7052-5.
Product Information
Cholesteryl-TEG Phosphoramidite (10-1975)
5'-Cholesteryl-TEG Phosphoramidite (10-1976)
α-Tocopherol-TEG Phosphoramidite (10-1977)
Stearyl Phosphoramidite (10-1979)
Glen-Pak™ is a trademark of Glen Research Corporation.
- Glen Report 24.11: Spermine Phosphoramidite: A Potent Modification with Many Applications
- Glen Report 24.12: New Product - S-Bz-Thiol-Modifier C6-dT
- Glen Report 24.13: New Products: AB 3900 Columns Containing 1000Å CPG
- Glen Report 24.14: New Product - Dibenzaocyclooctyl (DBCO) Copper-Free Click Chemistry
- Glen Report 24.15: New Products - 6-HEX and 6-TET Azides
- Glen Report 24.16: New Products - Solid, Stable Amino-Modifiers - PDA Amino-Modifiers
- Glen Report 24.17: New Product - 5'-Stearyl Phosphoramidite - An Alternative Lipophilic Carrier
- Glen Report 24.18: Technical Brief – Compatibility of UltraMild Monomers or Ac-dC with Hydrazine Reagent

