Glen Report 19.27: New Products - 2,6-Diaminopurine-TOM and Fmoc-Amino-Modifier C6 dT Amidites
2,6-Diaminopurine-TOM
We are happy to provide 2,6-Diaminopurine ribonucleoside (2-Amino-A) TOM-CE Phosphoramidite with a new protection scheme optimized for methylamine deprotection of RNA oligos with TOM-protecting groups. Under standard TOM deprotection conditions (10M MeNH2 in H2O/EtOH, RT), our previous diacetyl protected monomer showed significantly different rates of deprotection for the N2-acetyl and N6-acetyl protecting groups. The optimized protection scheme using N2-methoxyacetyl and N6-isobutyryl protecting groups was reported1 by Porcher and Pitsch.
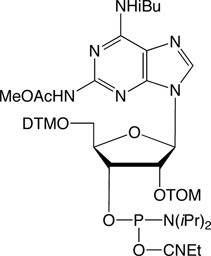
2,6-Diaminopurine-TOM
The following stabilities were determined for diacetyl protected diaminopurines:
N2,N6-diacetyl diaminopurine:
1/2 (N6) < 2 min and t1/2 (N2) = 30 min
The new protecting scheme with the more labile N2-methoxyacetyl and the more resistant N6-isobutyryl protecting groups showed the following deprotection rates:
N2-methoxyacetyl, N6-isobutyryl diaminopurine:
t1/2 (N6 and N2) = 3 min
By contrast, under the same conditions, N2-acetylguanosine, which is the most resistant to deprotection of the TOM-protected monomers, showed a similar deprotection rate:
N2-acetylguanosine: t1/2 = 4 min
Conclusion
The new protection scheme for 2,6-Diaminopurine ribonucleoside is virtually ideal for RNA synthesis and leads to simultaneous deprotection of both protecting groups of the diaminopurine, and the stability of the new monomer is comparable to the stability of N2-acetylated guanosine.
Reference
- S. Porcher and S. Pitsch, Helv Chim Acta, 2005, 88, 2683-2704.
Product Information
2,6-Diaminopurine-TOM-CE Phosphoramidite (10-3085)
Fmoc-Amino-Modifier C6 dT
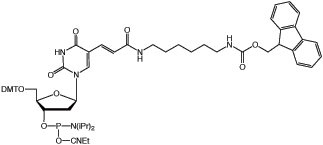
Fmoc-Amino-Modifier C6 dT
Amino-Modifier C6 dT is one of our oldest and best accepted products, and has become virtually a standard in oligonucleotide modification and labelling. Over the years, we have introduced dA, dC, dG and U variants and these products have also found favor as vehicles for highly specific labelling of oligonucleotides. However, these products are unable to address one need which our customers have requested - on-column labelling. In all of these cases, the amino group is protected with the trusty trifluoroacetate (TFA) protecting group which is intended to be removed during the standard oligonucleotide deprotection. Try as we might, we have never found conditions which allow the TFA group to be removed while the oligonucleotide remains attached to the support. Our initial attempts to solve this problem by using a 9-fluorenylmethoxycarbonyl (Fmoc) protecting group were not encouraging. However, a simple adjustment to the procedure used to remove the Fmoc protecting group allows the product to function properly. The problem was caused by alkylation of the amino group by acrylonitrile formed on elimination of the cyanoethyl phosphate protection groups. By using a two step procedure, the first to remove the cyanoethyl protection groups and flush the formed acrylonitrile from the synthesis column using 1% diisopropylamine in acetonitrile, and the second to remove the Fmoc group using 10% piperidine in DMF. The amino group so formed on the column can be reacted with a variety of activated esters. The one caveat is that the added tag must be stable to the basic conditions of the oligonucleotide cleavage and deprotection.
Product Information
- Glen Report 19.21: Glen-Pak™ Cartridges – The Ultimate Purification Cartridges
- Glen Report 19.22: Technical Brief - Procedure for the Synthesis and Deprotection of Synthetic RNA
- Glen Report 19.23: Improved RNA Synthesis Using the Q-Linker for Faster Product Cleavage from Solid-Phase Supports
- Glen Report 19.24: New Lines of Monomers - HT for High Throughput and LC for Low Cost RNA
- Glen Report 19.25: AP-dC is compatible with PCR and is Highly Fluorescent
- Glen Report 19.26: Pyrrolidine CE Phosphoramidite: A New Base Excision Repair Inhibitor
- Glen Report 19.27: New Products - 2,6-Diaminopurine-TOM and Fmoc-Amino-Modifier C6 dT Amidites
- Glen Report 19.28: Versatile New Reagents – Pyrene-dU and Perylene-dU
- Glen Report 19.29: Technical Brief - About Activators: Now and Tomorrow
- Glen Report 19.210: Introducing Thiophosphoramidites

