Glen Report 11.13: New Fluorescent Reagents - TAMRA-dT, Dabcyl-dT
At Glen Research, customer service extends beyond looking after our customers' orders. We also listen to our customers' needs and try to take every opportunity to streamline their production processes where possible. Especially in the area of fluorescent labelling, post-synthesis conjugation of dyes to amino- or thiol-modified oligonucleotides is inefficient and imposes a heavy purification burden to eliminate unconjugated excess dye. We are happy to introduce two new dT derivatives which streamline oligo production currently carried out by post-synthesis conjugation.
In our reviews of current literature, we have previously observed that product applications in the area of fluorescent labelling are extensively covered by patents. We would, therefore, request that our customers make themselves aware of any patents covering the processing of fluorescent oligonucleotides and the uses that might apply to their work.
TAMRA-dT
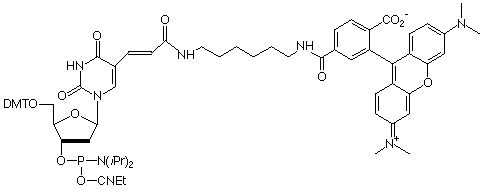
To supplement the tetramethylrhodamine (TAMRA) NHS ester and TAMRA-CPG we currently supply, we are offering TAMRA-dT for adding TAMRA within the oligo sequence. Because TAMRA is not stable to ammonium hydroxide, the procedure to cleave and deprotect the labelled oligonucleotide must be changed. We recommend using the UltraMILD monomers and deprotection with potassium carbonate in methanol. In this way, TAMRA oligonucleotides can be conveniently isolated.
A recent publication1 describes an alternative TAMRA support, along with a procedure2 to allow the use of regular monomers rather than our UltraMILD set. Cleavage and deprotection is carried out using t-butylamine/methanol/water (1:1:2).
Dabcyl-dT
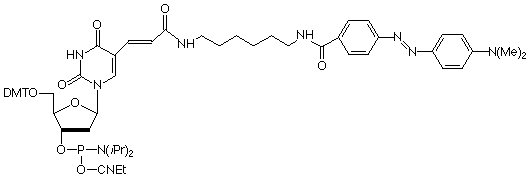
.
Molecular beacon probes3 have come to rely on the fluorescence quenching properties of the dabcyl molecule. A standard molecular beacon has a stem loop structure with a fluorophore like fluorescein at the 5'-terminus along with the quencher, usually a dabcyl group, at the 3'-terminus.
A further very elegant application of the quenching properties of the dabcyl group was described recently.4 Again the oligos contained the fluorophore at the 5'-terminus but the dabcyl group was located within the sequence. The structure of the primer should allow it to form a hairpin in such a way that the fluorophore is spatially adjacent to the quencher in the hairpin stem. The oligos can then be used as PCR primers. The fluorescence intensity of the amplified product correlates with the amount of incorporated primers since the hairpin no longer exists in the double-stranded amplified product. Indeed, the fluorescent signal is only present when the primers are incorporated into the double-stranded amplified product. The authors note that this technology eliminates the risk of carry-over contamination, simplifies the amplification assay, and opens up new possibilities for the real-time quantification of the amplified DNA over an extremely wide dynamic range.
The hairpin primers were produced by substituting an appropriate dT residue with Amino-Modifier C6-dT and incorporating fluorescein at the 5'-terminus with a 5'-fluorescein phosphoramidite. The dabcyl group was added post-synthetically by conjugating the Amino-Modifier C6-dT with dabcyl-NHS ester. We now simplify the production process by offering Dabcyl-dT phosphoramidite for direct incorporation into the quencher site.
References
B. Mullah and A. Andrus, Tetrahedron Lett, 1997, 38, 5751-5754.
S.L. Woo, S.M. Menchen, and S. Fung, 1993, US Patent No. 5,231,191.
S. Tyagi and F.R. Kramer, Nature Biotechnology, 1996, 14, 303-308.
I.A. Nazarenko, S.K. Bhatnagar, and R.J. Hohman, Nucleic Acids Res., 1997, 25, 2516-21.
Product Information
- Glen Report 11.11: Using Modified Bases to Optimize Hybridization
- Glen Report 11.12: Oligo Affinity Supports
- Glen Report 11.13: New Fluorescent Reagents - TAMRA-dT, Dabcyl-dT
- Glen Report 11.14: The Use of Puromycin CPG in Combinatorial Chemistry
- Glen Report 11.15: Update - Universal Supports, Q-supports
- Glen Report 11.16: More Novel Monomers - 6-Thio-dG, 2'-Amino-RNA, 5-Hydroxy-dC,dU Spacer C18, Spacer 9-CPG

