Glen Report 5-13: BioTEG - The Ultimate Biotin Phosphoramidite
At Glen Research, we have been continiuing our search for the ultimate biotin phosphoramidite for direct labelling of synthetic oligonucleotides. The follwing criteria have directed our efforts:
- The product must be soluable in acetonitrile at concentrations useful for DNA synthesis.
- The product must include a DMT group for cartridge purifications. This requirement is imperative in the design of biotinylated PCR primers beause of the potential for cross contamination in HPLC purifications.
- The product must also be capable of branching to allow multiple biotins to be introduced at the 3'- or 5'-terminius for probe develpment.
- Oligonucleotides or double stranded DNA labelled with this biotin phosphoramidite must be adequately captured by streptavidin magnetic beads.
A recent paper described1 a product which seemed to meet our design criteria. The product is soluble in acetonitrile and is capable of being inserted in multiple additions at the 3' or S' ter.minus. This product is similar in concept to our 5'-Branched Modifier C32 in that, if inserted in the oligonucleotide sequence, the internucleotide linkage contains 2 carbon atoms. (The natural 2'-deoxyribose linkage contains 3 carbons.) Our current biotin phosphoramidite produces a more natural internucleotide linkage containing 3 carbon atoms and has been used successfully and productively.
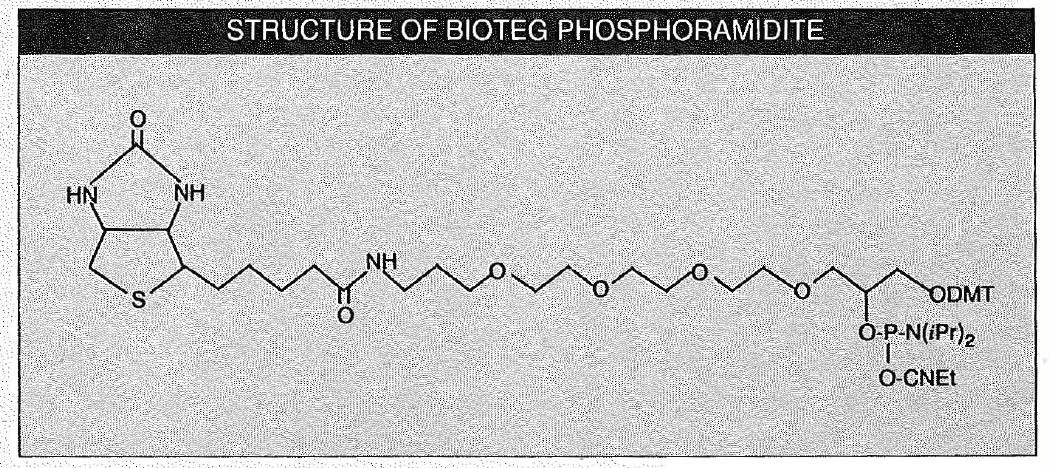
If inserted at the 5' terminus, the spacer arm of most commercially available biotin phosphoramidites, including our original product, consists of 6 atoms. There are indications that for some applications, diagnostic probes and capture of long, amplified DNA sequences, it is possible that a longer spacer arm would be desirable. A recent patent3 discloses functionalizing reagents useful for the introduction of thiol and amino groups into oligonucleotides using a long hydrophilic spacer arm. Using a similar strategy, we have designed and now introduce BioTEG Phosphoramidite, which contains a 15 atom mixed polarity spacer arm based on a triethylene glycol. This product would seem to meet all of our design criteria and may, in time, deserve to be referred to as the ultimate biotin phosphoramidite:
BioTEG phosphoramidite is now available in a variety of pack sizes to suit every need. Use a 50µmole pack to add a single biotin to the 5' terminis or one of the two larger packs for several additions. The product is used in a manner identical to normal nucleoside phosphoramidites with the exception that a longer coupling step (10-15 minutes) is needed to give high coupling efficiency.
For use at the 3'-terminus, we are happy to introduce BioTEG CPG. Again the use of this reagent is totally compatible with automated synthesizers.
References
1) K. Misiura, I. Durrant, M.R. Evans, and M.J. Gait, Nucleic Acids Res., 1990, 18, 4345:
2) P.S. Nelson, R, Sherman-Gold, and R. Leon, Nucleic Acids Res., 1989, 17, 7179.
3) C. Levenson, C. Chang, and F.T. Oakes, US Patent 4,914,210, 1990.
Product Information
- Glen Report 5-11: 2'-0Me-RNA -The Logical RNA Alternative?
- Glen Report 5-12: Psoralen C2 / Direct Fluorescein Labelling of Oligonucleotides
- Glen Report 5-13: BioTEG - The Ultimate Biotin Phosphoramidite
- Glen Report 5-14: Blocking the Termini of Oligonucleotides
- Glen Report 5-15: Thiol Modification of Oligonucleotides
- Glen Report 5-16: Spacer Phosphoramidite
- Glen Report 5-17: Unusual Monomers

