Glen Report 27.14: Photocleavable Biotin Linker for Use in SOMAscan™
Prepared by: Jeff Carter
Director, Process Chemistry
SomaLogic, Inc.
2945 Wilderness Pl., Boulder, CO 80301
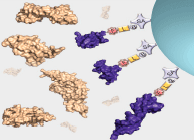
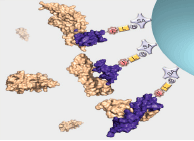
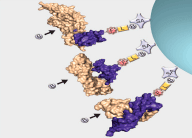
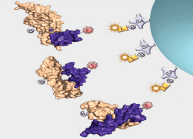
Interrogation of the human proteome in a highly multiplexed and efficient manner remains a coveted and challenging goal in biology. SomaLogic has implemented a new aptamer-based proteomic technology for biomarker discovery capable of simultaneously measuring thousands of proteins from small sample volumes (65 µL of serum or plasma). Our current assay allows us to measure >1000 proteins with very low limits of detection (300 fM median), 7 logs of overall dynamic range, and 5% average coefficient of variation1,2. This technology is enabled by a new generation of aptamers that contain chemically modified nucleotides, which greatly expand the physicochemical diversity of the large randomized nucleic acid libraries from which the aptamers are selected. Proteins in complex matrices such as plasma and serum are measured with a process that transforms a signature of protein concentrations into a corresponding DNA aptamer concentration signature, which is then quantified with a DNA microarray. In essence, this assay takes advantage of the dual nature of aptamers as both folded binding entities with defined shapes and unique sequences recognizable by specific complementary hybridization probes (see Figure 1).
For more than two decades, there has been growing interest in proteomic biomarker screening technologies1,3. Though several technologies have been applied to this effort with some limitations4,5, recent work by SomaLogic has demonstrated the use of slow off-rate modified aptamer (SOMAmer®) reagents to enable multiplexed screening of thousands of serum or plasma proteins. Using the SOMAscan™ assay, we are able to measure >1000 proteins and are working towards a 3000-plex. The use of the photocleavable biotin (PCB) reagent6,7 as a capture/release agent in our assay (see Figure 1) has enabled assay development and implementation for the use of these highly specific and diverse set of reagents. Integration of the PCB reagent and a suitable fluorophore (i.e. cyanine-3) onto the termini of SOMAmer reagents with suitable modified nucleotide positions2,8,9 (see Figure 2, Page 8) affords these measurements through binding of cognate proteins and subsequent quantification on a DNA microarray. Further work is underway to expand content and implement the larger 3000-plex with appropriate assay improvements and validation.





Side chain abbreviations: Bn, benzyl; Pe, 2-phenylethyl; Pp, 3-phenylpropyl; Th, 2-thiophenylmethyl; FBn, 4-fluorobenzyl; Nap, 1-naphthylmethyl; 2Nap, 2-naphthylmethyl; Ne, 1-naphthyl-2-ethyl; 2Ne, 2-naphthyl-2-ethyl; Trp, 3-indole-2-ethyl; Bt, 3-benzothiophenyl-2-ethyl; Bf, 3-benzofuranyl-2-ethyl; Bi, 1-benzimidazol-2-ethyl; Tyr, 4-hydroxyphenyl-2-ethyl; Pyr, 4-pyridinylmethyl; MBn, 3,4-methylenedioxybenzyl; MPe, 3,4-methylenedioxyphenyl-2-ethyl; 3MBn, 3-methoxybenzyl; 4MBn, 4-methoxybenzyl; 3,4MBn, 3,4-dimethoxybenzyl; RTHF, R-tetrahydrofuranylmethyl; STHF, S-tetrahydrofuranylmethyl; Moe, morpholino-2-ethyl; Thr, R-2-hydroxypropyl; iBu, iso-butyl
(Adapted from Rohloff et al, Molecular Therapy – Nucleic Acids, 2014, 3, e201)
References
- Gold et al, (2010), Aptamer-based multiplexed proteomic technology for biomarker discovery, Nature Precedings
- Rohloff et al, (2014), Nucleic Acid Ligands With Protein-like Side Chains: Modified Aptamers and Their Use as Diagnostic and Therapeutic Agents, Molecular Therapy – Nucleic Acid, 3, e201
- Zichi, D. et al, (2008), Proteomics and diagnostics: Let’s get specific, again. Curr. Opin. Chem. Biol. 12, 78-85
- Knezevic et al, (2001), Proteomic profiling of the cancer microenvironment by antibody arrays, Proteomics, 1, 1271–1278
- Gregorich and Ge,(2014), Top-down proteomics in health and disease: Challenges and opportunities, Proteomics, 14, 1195–1210
- Olejnik et al, (1996), Photocleavable biotin phosphoramidite for 5’-end-labeling, affinity purification and phosphorylation of synthetic oligonucleotides, Nucleic Acids Research, 24:2, 361-366
- Olejnik et al, (1999), Photocleavable peptide-DNA conjugates: synthesis and applications to DNA analysis using MALDI-MS, Nucleic Acids Research, 27:33, 4626-4631
- Vaught, J. D. et al, (2010), Expanding the chemistry of DNA for in vitro selection, J. Am. Chem. Soc. 132, 4141-4151
- Rohloff et al, (2015), Practical Synthesis of Cytidine-5-Carboxamide-Modified Nucleotide Reagents, Nucleosides, Nucleotides and Nucleic Acids, 34:3, 180-198
Also see: PC Modifiers
Product Information
- Glen Report 27.11: Reversible Photo-Switching of DNA Function with Azobenzene-Tethered DNA
- Glen Report 27.12: New Products - Dithiol Serinol Phosphoramidite and 3’- Dithiol Serinol CPG
- Glen Report 27.13: Technical Brief - APA: An Alternative to AMA Deprotection
- Glen Report 27.14: Photocleavable Biotin Linker for Use in SOMAscan™
- Glen Report 27.15: PC Modifiers
- Glen Report 27.16: Technical Brief - Capping and Trityl-Protected Amino-Modifiers
- Glen Report 27.17: Technical Brief - DBCO-dT - An Unusual Case of Iodine Sensitivity

