Glen Report 20.17: New Product - Deuterated 2’-Deoxyguanosine Phosphoramidite
For several decades, perdeuteration and selective deuteration have been useful approaches for the simplification of NMR spectra and for other structural studies of large biomolecules. Consequently, deuteration of nucleic acids and proteins has found widespread applications especially in multinuclear multidimensional solution NMR studies of these complex molecular systems.
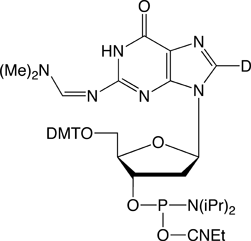
8-D-dmf-dG
Researchers have focused attention on the efficient syntheses of nucleosides and nucleotides containing deuterium (2H, D) labeled base residues. These reactions and the subsequent purification of the final products are easier to perform and much more cost-effective than multi-step chemical and biochemical procedures for sugar deuteration. According to Huang1, in typical two dimensional (2D) NOESY spectra of oligonucleotides, structural information is primarily contained in the region that connects base with sugar protons (as shown in Figure 2). If the selected base protons, especially H8 in purines and H6 in pyrimidines, are replaced with 2H, all the pertinent cross peaks correlating the base to sugar protons will disappear.
Also, according to Chirakul2, many reported structures represent ground states that may bear little resemblance to the structures of the transition states. Therefore, structural information about ground states needs to be augmented with knowledge about the movements of functionally important residues. Solid state 2H NMR is a valuable tool to study motions of specific sites in nucleic acids. Researchers who are interested in studying the role of dynamics in the interaction of proteins with DNA by solid state 2H NMR require site-specific incorporation of deuterium-labeled nucleotides. To study the base motion of dA in DNA, Chirakul2 incorporated 2H into the 2- or 8-position of the adenine base.
We are happy to offer the first commercially available 2H modified phosphoramidite, 8-deutero-2’-deoxyGuanosine phosphoramidite (8-D-dmf-dG) (Figure1), to our customers involved in NMR spectroscopy. This phosphoramidite can be used on any DNA synthesizer using standard protocols. However, it is necessary to deprotect oligos containing 8-D-dmf-dG with 25% deuterated ammonium hydroxide (ND4OD) for 40 hours at room temperature to prevent deuterium exchange.1
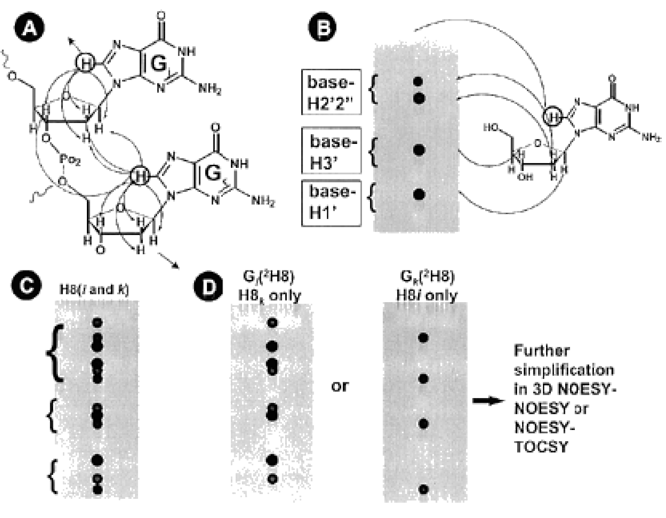
Schematic illustration of the effect of base 2H-labeling in NOESY spectrum.
(A) The conventional intra- and inter-residue connectivities in a right handed helix of nucleic acids.
(B) The corresponding 2D NOE connectivities between the protons of a base and a sugar residue and
(C) An ideal NOESY spectrum in the base to sugar proton region with the two base proton resonances superimposed.
(D) The predicted simplified NOESY spectra using site-specifically 2H-labeled samples.
(Illustration Courtesy of Reference 1)
References
- X. Huang, P. Yu, E. LeProust, and X. Gao, Nucleic Acids Res., 1997, 25, 4758-4763.
- P. Chirakul, J.R. Litzer, and S.T. Sigurdsson, Nucleosides, Nucleotides and Nucleic Acids, 2001, 20, 1903-1913.
Ordering Information
8-D-dmf-dG-CE Phosphoramidite has been discontinued.
- Glen Report 20.11: An Unnatural Base Pair System for the Expansion of Genetic Information
- Glen Report 20.12: Thiophosphoramidites and Their Use in Synthesizing Oligonucleotide Phosphorodithioate Linkages
- Glen Report 20.13: Technical Brief - Purification of 6-FAM Labelled oligos using Glen-Pak™ Cartridges
- Glen Report 20.14: More Click Chemistry
- Glen Report 20.15: Technical Brief - New application of 5-Me-iso-dC and iso-dG
- Glen Report 20.16: Technical Brief - New application for 5’-OMe-dT phosphoramidite
- Glen Report 20.17: New Product - Deuterated 2’-Deoxyguanosine Phosphoramidite
- Glen Report 20.18: New Products - Solid CPR II / Formylindole – Aldehyde Modifier
- Glen Report 20.19:New Product - 5'-Cholesteryl-TEG Phosphoramidite
- Glen Report 20.110: Improving Universal Support II for Oligonucleotide Synthesis
- Glen Report 20.111: Differences Between Universal Support II and III
- Glen Report 20.112: Technical Brief - Phage Display, Artificial Antibodies and Trimer Phosphoramidites

