Glen Report 32-22: New Products — AquaPhluor® 639 5’-Phosphoramidite and CPG
New Products — AquaPhluor® 639 5’-Phosphoramidite and CPG
AquaPhluor® 639 (AP639) is a rhodamine dye with an excitation and emission in the far red of the visible spectrum. Structurally, it closely resembles the non-Cyanine AquaPhluor® 593 we introduced a few years back, but functionally, it is an alternative to Cyanine 5. As described in the previous article, AP639 has several advantages over Cyanine 5, and in collaboration with ELITechGroup, Glen Research is pleased to offer the phosphoramidite and CPG versions of this dye for 5’ or 3’ labeling, respectively (Figure 1).
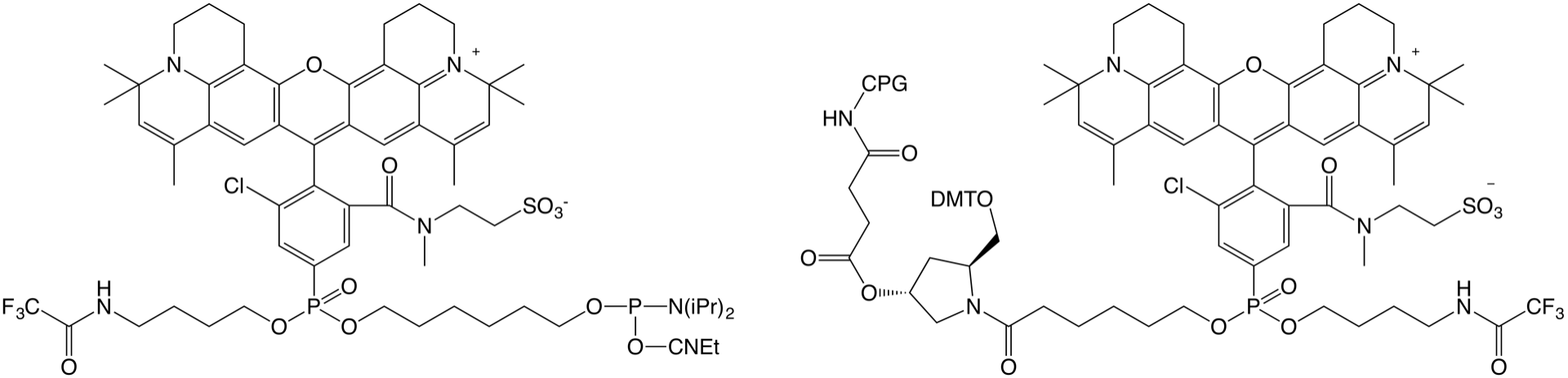 |
|
| 5'-AquaPhluor® 639 Phosphoramidite | 5'-AquaPhluor® 639 CPG |
Figure 1. New AquaPhluor® 639 Products |
|
The use of these AP639 products is relatively straightforward. The phosphoramidite can be incorporated with a 3-minute coupling time, and the support should be used in the same way a normal protected nucleoside support would, as it contains a DMT group. For deprotection, most standard conditions are generally compatible.
In addition to having photostability and temperature-independent spectral properties superior to those of Cyanine 5, AP639 shows highly improved chemical stability under basic conditions. Those who have experience with Cyanine 5 will know that it must be deprotected gently. In the presence of standard deprotection conditions, Cyanine 5 will slowly go from a deep blue, to green, to eventually yellow. UltraMild deprotection is ideal, but ammonium hydroxide at room temperature for up to 36 hours will also provide acceptable amounts of intact Cyanine 5. On the other hand, deprotection stability is generally not an issue for AP639. In fact, it is the opposite of Cyanine 5, as UltraMild deprotection will not suffice in removing the AP639 TFA-aminobutyl group. Deprotection for AP639 was evaluated in a range of different conditions (Table 1), and all were suitable. In more aggressive deprotection conditions, an extra non-fluorescent peak or two may be present on the baseline in addition to the major desired product, but these are all minor in intensity and not unusual for most dye-labeled oligonucleotides. For example, with AMA, we observed a small non-fluorescent, more hydrophobic peak by RP-HPLC. According to ESI-MS, it matches the displacement of the N-methyl taurine amide by methylamine, and the absence of fluorescence is likely due to the formation of an additional lactam ring. The excellent chemical stability of AP639 should prove particularly useful in highly modified oligonucleotides for which mild deprotection methods are not possible.
| Solution | Temperature (°C) | Time (h) |
| NH4OH | RT | 17 |
| 55 | 17 | |
| 65 | 2 | |
| AMA | 65 | 0.17 (10 min) |
| tBuNH2/H2O 1:3 | 60 | 6 |
IP Statement
This Product is for research purposes only, and may not be used for commercial, clinical, diagnostic or any other use. The Product is subject to proprietary rights of ELITechGroup and is made and sold under license from ELITechGroup. There is no implied license for commercial use with respect to this Product and a license must be obtained directly from ELITechGroup. With respect to any proposed commercial use of this Product, "commercial use" includes but is not limited to the sale, lease, license or other transfer of the Product or any material derived or produced from it, the sale, lease, license, or other grant of rights to use the Product or any material derived or produced from it, or the use of the Product to perform services for a fee for third parties (including contract research). AquaPhluor®, Yakima Yellow®, Redmond Red® and Eclipse® are registered trademarks of ELITechGroup.
Product Information
- Glen Report 32-21: AquaPhluor® 639
- Glen Report 32-22: New Products — AquaPhluor® 639 5’-Phosphoramidite and CPG
- Glen Report 32-23: New Products - ÄKTA Capping Reagents
- Glen Report 32-24: Very Fast CRISPR (vfCRISPR) “On Demand”
- Glen Report 32-25: Application Note — NPOM-Caged-dT
- Glen Report 32-26: Technical Brief - NHS Esters
- Glen Report 32-27: Technical Snippets

