Glen Report 12.13: Poly-Pak Purification of Labelled Probes
Introduction
The demand for fluorescently labelled oligonucleotides has been steadily increasing. With the advent of fluorophores supplied as both CPG and phosphoramidites, oligo synthesis has become routine and efficient. Yet their purification remains a bottleneck in production, due primarily to the failure sequences being more hydrophobic than the full length oligo when a 3'-dye is present. What is needed is a means by which the oligo can be first purified by length, and then afterwards, by means of the dyes to remove any unlabeled sequences. A new Poly-Pak purification protocol developed at Glen Research allows one to do exactly this.
Protocols are tailored to specific dye combinations such as those seen in FRET or Molecular Beacon probes. The strategy behind the purification lies in washing the Poly-Pak cartridge with acetonitrile solutions at low and neutral pH. After first loading the oligo on to the cartridge, the phosphodiester backbone is protonated using 2% TFA. Then a rinse is performed with an acetonitrile/2% TFA solution (Solution A). With the phosphate backbone protonated, the hydrophobicity of the oligo increases as a function of length and so, the failure sequences are selectively removed without loss of full length product. Then the pH is brought back to neutrality and a second wash with an acetonitrile/TEAA solution (Solution B) is performed. At pH 7, the presence of the dyes is the deciding factor in the oligo's affinity to the Poly-Pak support and any sequences without the dyes present, or intact, are removed.
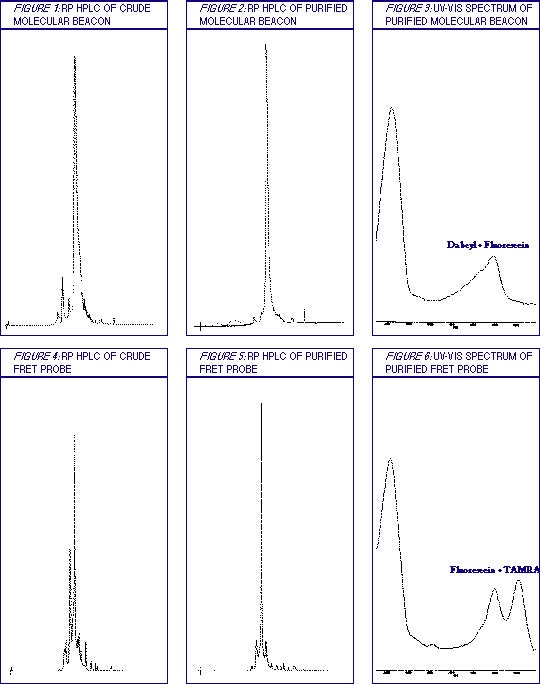
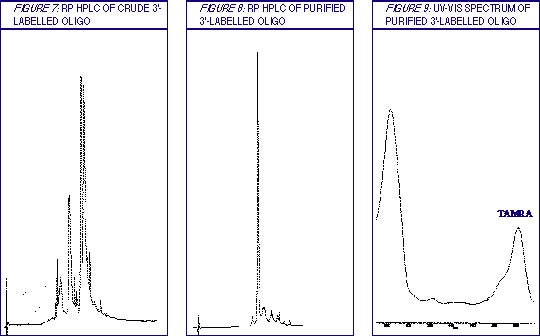
Table 1 contains a general description of the procedure, while details of the wash Solutions A and B are collected in Table 2. Examples of the purification results are shown in Figures 1-9.
Cartridge Preparation
The flow rate of solvents through the Poly-Pak cartridge should be dropwise.
- 1. Connect a syringe to the female luer of the cartridge and have the male luer terminate in a waste vessel.
- 2. Flush the cartridge with 4 mL acetonitrile followed by 4 mL 2 M TEAA.
Sample Preparation
- 3. Following synthesis, deprotect the oligo. If 50 mM K2CO3 in anhydrous methanol has been used, continue to the next step. If ammonia or AMA was used for deprotection, dry the probe down, take up in 1 mL dH2O and then continue with the following step.
- 4. Add 1 volume 2 M TEAA followed by 8 volumes of deionized water.
Purification Procedure
- 5. Load the sample solution onto the cartridge . Reload if necessary after addition of a few drops of 2 M TEAA which increases the affinity of the oligo for the support.
- 6. Flush cartridge with 5 mL 2% TFA; do this whether the oligo is DMT ON or OFF.
- 7. Rinse cartridge with 15 mL of Solution A.
- 8. Flush cartridge with 4 mL dH2O.
- 9. Rinse cartridge with 10 mL of Solution B.
- 10. Elute product with 3-4 mL of 50% Acetonitrile in Water.
Notes
The percent ACN used in Solution A rinse depends mostly upon the oligo length, though the type and number of dyes present also is a factor. In terms of length, a rule of thumb is to increase the percent ACN by 0.5% per base added. The percent ACN in Solution B is most strongly dependent upon the hydrophobicity of dye(s) labelled on the oligo and is independent of oligo length. An oligo with two very hydrophobic dyes such as Cy5 and Dabcyl can require up to 25% ACN in 0.1 M TEAA.
Finally, if the oligo does not bind quantitatively to the cartridge during loading, lower the pH by adding a few drops of glacial acetic acid. Conversely, if there is difficulty getting all the purified oligo off the cartridge when eluting, add a drop of 30% ammonia to the 50% ACN.
Some notes on individual probe types follow:
Molecular Beacons
This protocol has been optimized using molecular beacons of 30 nucleotides in length that contain either fluorescein or Cy5 at the 5' terminus and Dabcyl on the 3' end. Using the Poly-PakTM II cartridge, 0.5 µmole of crude oligo (approximately 50 OD units) may be purified; overloading the cartridge results in lower probe yield and purity. Due to the difference in hydrophobicity of the Cy5 and fluorescein, different rinsing solutions are used. For beacons that contain fluorophores of intermediate hydrophobicity, e.g., TAMRA, try an intermediate concentration of ACN in the TEAA buffer for Solution B.
FRET Probes
This protocol has been optimized for 16-20mers containing both Fluorescein and TAMRA on a 0.5 µmol scale (approximately 50 OD units); overloading the cartridge results in lower probe yield and purity. Other dye combinations may require more stringent wash conditions depending upon the hydrophobicity of the dyes. Tetramethylrhodamine (TAMRA) dyes are sensitive to ammonia and will degrade in its presence; use UltraMILD DNA monomers (see 'UltraMild DNA Synthesis' in the Glen Research catalog) and deprotect in 50 mM K2CO3 in anhydrous methanol for 4 hours at room temperature. Using 0.02 M I2 for phosphite oxidation will lead to a cleaner probe.
3'-Dye-labelled Oligonucleotides
This protocol has been optimized for 20mers labeled with either TAMRA or fluorescein on the 3' terminus. The loading used was 50-60 OD per Poly-Pak cartridge (approximately 0.5 µmole oligonucleotide). For longer oligos, or oligos that are labelled with a more hydrophobic dye, e.g., Dabcyl, use a higher ratio of ACN to 2% TFA in Solution A.
Product Information
Poly-Pak™ Reverse Phase Purification Products have been discontinued
- Glen Report 12.11: Oligonucleotide Dendrimers: From Poly-Labelled DNA Probes to Stable Nano-Structures
- Glen Report 12.12: TOM-Protected Minor Base RNA Phosphoramidites
- Glen Report 12.13: Poly-Pak Purification of Labelled Probes
- Glen Report 12.14: MerMade Instruments
- Glen Report 12.15: More Novel Monomers: Nucleoside α-Thiotriphosphates, 5-Hydroxy-Methyl-dU, Dabcyl Products, and a Highly Fluorescent Nucleoside
- Glen Report 12.16: Product Update - Which Chemical Phosphorylation Reagent?

