Glen Report 26.13: New Product - 5-Me-dC Now Compatible with AMA and UltraMild Deprotection
One of the earliest minor bases introduced by Glen Research was 5-Me-dC-CE Phosphoramidite. 5-Me-dC has always been interesting to researchers because it stabilizes duplexes relative to dC by 1.5° per insertion. This desirable effect is attributed to the hydrophobic nature of the C5 methyl group which helps exclude water from the duplex. Antisense and diagnostic developers have long made use of 5-Me-dC as a simple and relatively inexpensive substitution for dC.
More recently, 5-Me-dC has generated interest in its own right in the burgeoning field of epigenetics. Methylation of dC usually occurs in areas with high concentration of CG bases (CpG islands) and the transformation of dC to 5-Me-dC is mediated by specialized DNA methyltransferase (DNMT) enzymes. Once methylated, 5-Me-dC can then be oxidized to form, consecutively, 5-hydroxymethyl-dC, 5-formyl-dC, and 5-carboxy-dC, all modified nucleosides of immense interest to epigenetics researchers.
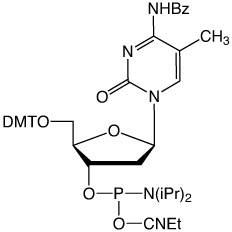
Our existing 5-Me-dC monomer (1) with the N4-amine protected with a benzoyl group has served the research community well for decades. However, this monomer is not compatible with deprotection using AMA. As shown in Figure 2, deprotection of a simple oligo containing benzoyl protected 5-Me-dC leads to around 7% of the N4-Me mutation caused by displacement of benzamide by methylamine. In addition, the benzoyl protecting group is not compatible with UltraMild deprotection since it is not removed by potassium carbonate in methanol.
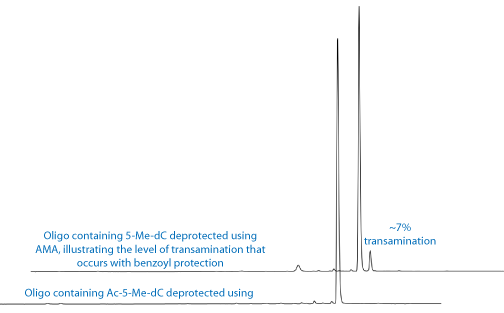
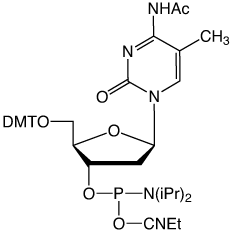
To remedy these shortcomings, we are introducing the acetyl-protected 5-Me-dC monomer, Ac-5-Me-dC-CE Phosphoramidite (2). This monomer is fully compatible with AMA deprotection and none of the N4-Me mutation is observed on deprotection, as shown in Figure 2. The N4-acetyl protecting group is also completely removed under the conditions of UltraMild deprotection.
We are happy to offer the acetyl-protected monomer in addition to its older benzoyl-protected cousin.
Historically, we have offered a CPG support for the benzoyl-protected 5-Me-dC. However, the acetyl version is fully compatible with AMA deprotection and, therefore, a universal support like Glen UnySupport can be used.
Product Information
- Glen Report 26.11: DNA conjugation to HaloTagged Protein Using Glen Bromohexyl Linker
- Glen Report 26.12: Preparation of Bromohexyl-Modified Oligonucleotides for HaloTag Conjugation
- Glen Report 26.13: New Product - 5-Me-dC Now Compatible with AMA and UltraMild Deprotection
- Glen Report 26.14: Technical Brief - AMA – Ubiquitous Reagent for Oligo Deprotection
- Glen Report 26.15: New Product - THPTA - A Water Soluble Click Ligand

