Glen Report 16.12: DTPA - Immobilize Oligos to Gold Surfaces by Multiple Thiol Anchorages
We are grateful to Dr. Peter Frischmann of FRIZ Biochem in Munich for providing us with the following information.
A prerequisite for reproducible and reliable results on DNA-arrays on gold substrates is an optimized immobilization chemistry. Although the gold-sulfur bond (with a bond energy of 30–40 kcal/mol) is a relatively strong anchor between a surface and biopolymer, it is well known that monofunctional thiolated oligonucleotides are slowly displaced from the gold surface at temperatures between 60–90°C and at high salt concentration buffers (1 M NaCl). Especially drastic effects are detected with biological buffer systems containing dithiothreitol or mercaptoethanol, which lead to complete displacement of the oligonucleotides. For DNA-arrays on gold substrates, it is critical to guarantee the stability of the anchor during the washing steps (mechanical stress). In addition, the lateral diffusion of monofunctional anchored molecules on gold surfaces has to be prevented.
A special reagent for multiple anchorage:
In order to immobilize biopolymers on gold surfaces, a stable anchor is essential to avoid decomposition. FRIZ Biochem has developed a new compound with a performance far superior to commercially available components. The stable immobilization of oligonucleotides on DNA-arrays is a prerequisite for reliable and reproducible results. By modifying the capture probes with this new compound, FRIZ's research has detected no loss of oligonucleotides for the range of conditions used for array technologies (i.e., mechanical stress caused by washing or hybridization procedures). This increased stability is the basis for robust DNA-chips on gold substrates.
Superior performance with Dithiol-Phosphoramidite(DTPA)
FRIZ Biochem's proprietary DTPA is a disulfide-containing phosphoramidite to modify synthetic DNA or RNA with multiple thiol groups (Figure 1). This novel product can be incorporated at any position of the oligonucleotide. The number of monomers, each forming two gold sulfur bonds, can be adapted to the demands of the application. The stability of the oligonucleotides tethered to the surface is increased by the multiple gold-anchors formed. Three units of the Disulfide-Modifier have proved to offer optimal stability, where the bonding is stressed by washing steps. Of course, this modifier can also be used for multiple reactions with maleimides and other thiol-specific derivatives.
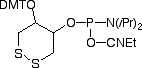
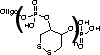
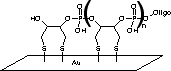
Stability tests of immobilized oligonucleotides on gold
The stability of thiolanchored oligo-nucleotides has been determined through fluorophore labelling studies. In FRIZ's experiments, commercially available monothiol modified oligonucleotides were compared with oligonucleotides immobilized with this new Disulfide-Modifier. A fluorophore (fluorescein) was attached at the 5' end of each oligonucleotide in order to study the immobilization and the stability with a fluorescence scanner. After the spotting of the oligonucleotides and washing with deionized water, the fluorescence intensities are shown in Figure 2 (green columns). To improve the performance of a bioassay, exposed bare gold surfaces should be avoided. In this case, FRIZ treated the surface with 1 mM mercaptohexanol. During this process, immobilized oligonucleotides are displaced according to the stability of their anchorage on the surface. The yellow columns in Figure 2 show the fluorescence intensities after this exposure. Up to 90% of oligonucleotides with one thiol modification were displaced from the gold. Under these conditions, the incorporation of three Disulfide-Modifiers (six gold-sulfur bonds) was shown to result in a stable bond – no oligonucleotides were displaced.
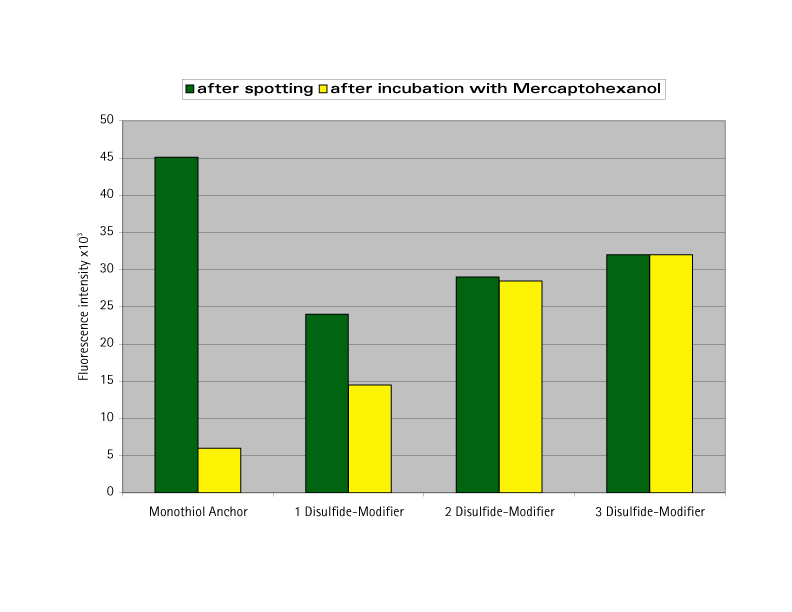
Fluorescence intensities of oligonucleotides anchored on a gold surface by different immobilization groups before and after mercaptohexanol (1mM) treatment.
DTPA is offered by Glen Research under license from FRIZ Biochem.
Product Information
Dithiol Phosphoramidite (DTPA) has been discontinued.
- Glen Report 16.11: Pyrrolo-C - A Fluorescent Nucleoside Base Analogue That Codes Efficiently as C
- Glen Report 16.12: DTPA - Immobilize Oligos to Gold Surfaces by Multiple Thiol Anchorages
- Glen Report 16.13: Minor Base and Related Novel Phosphoramidites
- Glen Report 16.14: 2'-Se-Uridine Phosphoramidite for RNA and DNA Derivatization in X-ray Crystallography

