AM Biotechnologies
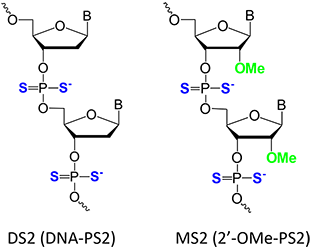
Advances in phosphorodithioate oligodeoxynucleotide (Figure 1: DNA-PS2 or DS2) chemistry, biochemical and biological studies have shown very interesting and potentially useful results. These include observations that DNA-PS2 or DS2 activates RNase H in vitro, strongly inhibits human immunodeficiency virus (HIV) reverse transcriptase, induces B-cell proliferation and differentiation, is completely resistant to hydrolysis by various nucleases, and binds to protein targets in the form of dithiophosphate aptamers (thioaptamers) (reviewed by Dr. Yang (1)). All these studies encouraged us to introduce the DNA-thiophosphoramidites to synthesize a DNA-PS2 or DS2 (Figure 1) several years ago.
Recently, siRNAs containing PS2 modifications were shown to exhibit favorable properties for therapeutic applications (2-4). Combining the PS2 with the 2’-O-methyl ribose modification (2’-OMe-PS2 or MS2 in Figure 1) afforded increased loading of modified siRNA duplexes into the RNA induced silencing complex (RISC) as well as enhanced gene-silencing antitumor activity (5). Interestingly, the former property can be attributed to MS2 modification at two residues adjacent to the 3’-TT overhang in the sense siRNA strand. This conclusion is supported by higher association based on in vitro pull-down assays, and tighter intracellular binding between MS2-siRNA and Ago2 protein, relative to the corresponding RNA and RNA-PS2. Very recently we have found that the use of MS2 substitution on a single nucleotide of 2’-OMe-RNA aptamer targeting VEGF-165 dramatically improves target binding affinity by approximately a thousand fold (from nanomolar to picomolar). In addition, the MS2-modified RNA aptamers demonstrate improved biochemical and biophysical characteristics (N. Dinuka Abeydeera et al. submitted to Nature communication). Moreover the X-ray crystal structures demonstrated that single PS2 or MS2 modified RNAs have virtually identical conformations with native RNA duplexes (6). In particular PS2 or MS2 modification does not trigger any changes in the ribose conformation and the backbone torsion angles (6). The structures of the PS2 and MS2 monomers are shown in Figure 4 on Page 5.
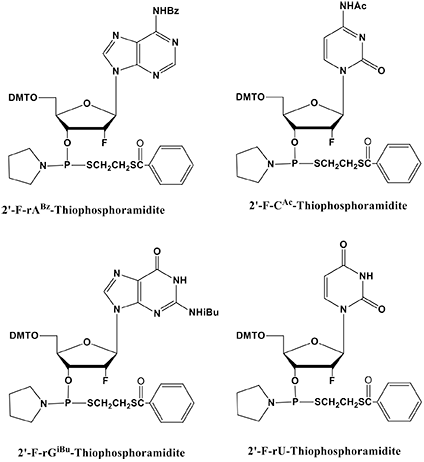
2’-Deoxy-2’-fluoro-RNA (2’-F-RNA) has been popular in ribozymes, siRNA, and anti-miRNA research. As a substitute for 2’-F-RNA phosphoromonothioate, we will make the four 2’-F-thiophosphoramidites available soon (Figure 3). In this way, we hope to open up a full spectrum of research applications for phosphorodithioates (PS2).
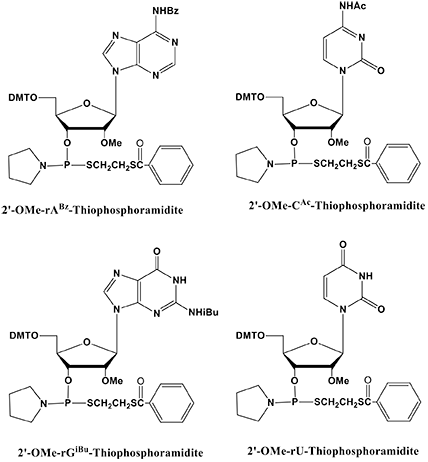
At this time, Glen Research is pleased to offer the 2'-OMe Thiophosphoramidites as shown in Figure 4.
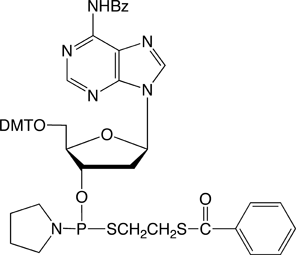
dA-Thiophosphoramidite |
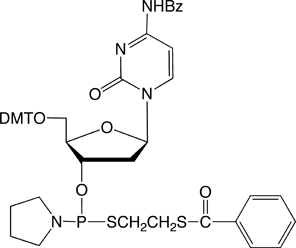
dC-Thiophosphoramidite |
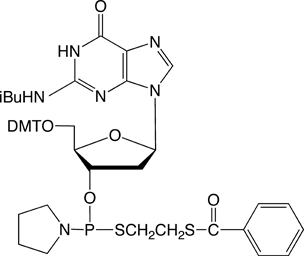
dG-Thiophosphoramidite |
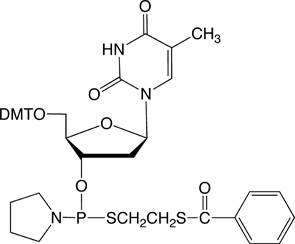
dT-Thiophosphoramidite |

2'-OMe-dA-Thiophosphoramidite |

2'-OMe-dC-Thiophosphoramidite |

2'-OMe-dG-Thiophosphoramidite |

2'-OMe-dT-Thiophosphoramidite |
Figure 4: The structures of thiophosphoramidites |
|