Glen Research is happy to confirm our agreement with Epoch Biosciences, Inc., announced on February 6, 2002, to sell several of Epoch's proprietary products designed for the synthesis of novel DNA probes. Initially we will provide products based on Epoch's Redmond Red™ and Yakima Yellow™ fluorophores and Eclipse™ non-fluorescent quencher, which will be described in detail in this article. We will also supply PPG, a modified nucleoside, which is covered in the article on Page 8 of this newsletter. It is a pleasure for us to expand our relationship with Epoch by helping to provide broad access for these compounds to research markets worldwide.
As part of the preamble to this article, it is instructive to quote from the Epoch press release, which positions these products perfectly. They note that the products covered by this agreement are as follows:
"all are innovative components for DNA probes, the workhorses of genetic analysis. Probes hybridize or bind to target DNA and then provide a signal so that the target can be detected. Many probes also carry a quencher that masks the signal until the binding has occurred. Redmond Red and Yakima Yellow are fluorescent dye tags that can be incorporated into synthetic DNA molecules to allow probe detection on a variety of platforms. The Eclipse Quencher is a new non-fluorescent quencher that allows DNA detection probes to be used for real time PCR applications such as measuring gene expression or detecting single nucleotide polymorphisms (SNPs). The PPG modified base is the first in a series of modified bases that Epoch has developed that allow DNA probes to be designed for regions of genomes that cannot be interrogated by probes containing normal nucleotide bases.

The use of fluorescent tags as an alternative to radiolabels in DNA probes and primers has blossomed over the years. Fluorescence is safely measured with inexpensive instrumentation and it is very straightforward to multiplex assays for exceptionally high throughput. Molecular beacon and fluorescence resonance energy transfer (FRET) probes can be used in assays, which can be carried out in closed tube formats with less sample handling at higher throughput. These probes are also suitable for use in techniques that include amplification of the target DNA.
Molecular beacon and FRET probes require efficient quenching until the probe is hybridized to the target. Molecular beacon probes are hairpin structures wherein the fluorescence is quenched by the proximity of the fluorophore to the quencher molecule. When the probe hybridizes to the target, it becomes linear, quenching is disrupted, and the probe fluoresces ready for detection. In FRET assays, when the probe is hybridized to the target, it is digested by nuclease activity in the polymerase being used for amplification of target copies. The fluorophore, released from the target and separated from the quencher, is now highly fluorescent and ready for detection. Fluorophore/quencher pairs can be chosen based on spectral properties — the emission of the fluorophore should overlap the absorption of the quencher. The quencher may absorb its partner's fluorescence and emit the fluorescence at a new wavelength or, in the case of a non-fluorescent quencher, as heat.
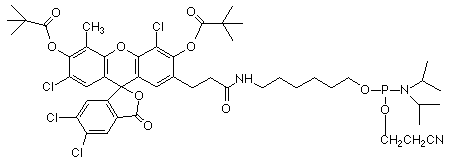
The new Epoch products offer two new fluorescent dyes, available immediately as phosphoramidites and supports, as shown in Figure 1. Their absorbance and emission characteristics are shown in Figure 2. Yakima Yellow has an absorption maximum at 530 nm and emission maximum at 549 nm, while Redmond Red's absorption and emission maxima are at 579 nm and 595 nm, respectively.






| Dye | λmax Abs | λmax Em | Extinc. at max | QY |
|---|---|---|---|---|
| Yakima Yellow | 530.5 nm | 549 nm | 84,000 | 0.96 |
| Redmond Red | 579 nm | 595 nm | 52,300 (pH 7.1) | 0.84 |
| 74,000 (pH 9.1) | ||||
|
Measured in 0.1 M sodium phosphate. As indicated, Redmond Red is pH sensitive. Yakima Yellow is not and has the same extinction coefficient at pH 7 as pH 9. The QYs (quantum yield of fluorescence) were determined in 0.1 M Tris-HCl pH 7.4 relative to reference dyes using the dye-labeled T12 oligo. For Yakima Yellow, the reference dye was carboxy tetramethylrhodamine in methanol. For Redmond Red, the reference dye was sulforhodamine 101 in EtOH. |
||||
Over the years, the dabcyl molecule has proved to be an excellent universal non-fluorescent (dark) quencher for molecular beacon probes. In addition, dabcyl is a very stable molecule and synthesis of doubly-labelled probes containing a dabcyl quencher is quite straightforward. The mechanism of quenching relies on the close proximity of the fluorophore to the dabcyl group, generally called static quenching, which is independent of spectral overlap between fluorophore and quencher. However in FRET probes, dabcyl's ability to act as a dark quencher is limited by its absorption spectrum to use with dyes emitting at 400 — 550 nm. Other fluorescent quenchers can be used to cover a broad spectrum of dyes, but synthesis of the doubly-labelled probes is made much more difficult by the lack of stability of some of the potential candidates to the conditions of oligo deprotection. These probes are best prepared in a two-step process requiring post-synthesis conjugation of the second dye, usually with purification problems.
The Eclipse Quencher from Epoch solves most of the problems inherent in the synthesis of molecular beacon and FRET probes. The Eclipse molecule is highly stable and can be used safely in all common oligo deprotection schemes. The absorption maximum for Eclipse Quencher is at 522 nm, compared to 479 nm for dabcyl. In addition, the structure of the Eclipse Quencher (Figure 1) is substantially more electron deficient than that of dabcyl and this leads to better quenching over a wider range of dyes, especially those with emission maxima at longer wavelength (red shifted) such as Redmond Red and Cy5. A simple experiment, using a molecular beacon probe, showed that quenching of the fluorescence of Cy5 was 53% more efficient with Eclipse Quencher than with dabcyl. Epoch researchers conducted a comparison1 of the quenching ability of the Eclipse Quencher relative to dabcyl in an enzymatic digestion assay designed to simulate the efficiency of quenching of FRET probes. Over the range of fluorophores from FAM (520 nm) to Cy5 (670 nm), Eclipse Quencher outperformed dabcyl in a measurement of signal to background ratio (signal, defined as fluorescence after digestion, divided by background, defined as initial fluorescence). The improvement was most marked for the higher wavelength dyes. In addition, with an absorption range from 390 nm to 625 nm (Figure 3), the Eclipse Quencher is capable of effective performance in a wide range of colored FRET probes.
As is normal these days, the use of these products is restricted and users should heed the following qualification statement:
"These Products are for research purposes only, and may not be used for commercial, clinical, diagnostic or any other use. The Products are subject to proprietary rights of Epoch Biosciences, Inc. and are made and sold under license from Epoch Biosciences, Inc. There is no implied license for commercial use with respect to the Products and a license must be obtained directly from Epoch Biosciences, Inc. with respect to any proposed commercial use of the Products. 'Commercial use' includes but is not limited to the sale, lease, license or other transfer of the Products or any material derived or produced from them, the sale, lease, license or other grant of rights to use the Products or any material derived or produced from them, or the use of the Products to perform services for a fee for third parties (including contract research)."
Redmond Red, Yakima Yellow, and Eclipse are trademarks of Epoch Biosciences, Inc.
